
BACKGROUND
Ligneous Conjunctivitis (LC) is the most common clinical manifestation of type-1 plasminogen (PLG) deficiency, a very rare autosomal recessive disorder with an estimated prevalence of ~1.6/million. LC is a membranous conjunctivitis characterized by the formation of fibrin-rich, soft/hard pseudomembranes on tarsal conjunctivae, resulting in visual impairment/loss, and commonly presents in the first weeks/months of life, but may present in childhood or any age. PLG is the precursor of plasmin; absence of plasmin activity results in impaired fibrinolysis and formation of fibrin-rich membranes.
LC affects bilateral eyes in up to 51% of cases, and corneal involvement, leading to blindness, occurs in 20-30% of cases. Although non-specific therapies have been reported to result in lesion improvement/resolution, only FFP administered topically or systemically, and topical/systemic plasminogen have demonstrated more consistent efficacy. Surgery is often performed for hard membrane removal; surgical intervention can trigger regrowth without adequate medical therapy. We report the long-term results of topical ocular administration from a PLG eye drop formulation [Kedrion Biopharma, CVP, Barga, Italy] for LC.
METHODS
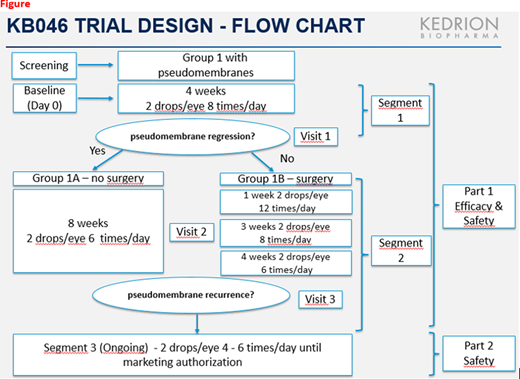
A phase 2/3, 3-segment [Fig], open label, historically controlled, clinical trial (KB046) was initiated in May 2013 at three sites (1 US; 2 Italy). Segments 1 & 2 assessed the efficacy and safety of short-term PLG treatment in pseudomembrane regression/recurrence and prevention. Subjects with residual hard membranes at Segment 1 end underwent surgical removal. Patients completing Segment 2 without pseudomembranes continued to segment 3 for long-term safety continuation. Segment 3 is ongoing.
Primary and secondary Efficacy Endpoints: a) Pseudomembrane recurrence prevention at end of Segment 2 and b) Regression of surface area of existing pseudomembranes at end of Segment 1 including Time (days) to pseudomembrane recurrence post-surgery or complete regression at end Segment 2. Regression and recurrence occurring during the trial were compared to the patients' prior treatments and surgical history. Safety Endpoints of segment 1/2 included overall safety, local tolerability, immunogenicity and viral safety.
RESULTS
Eleven patients (15 eyes) with pseudomembranes were enrolled in segment 1 and treated for 4 weeks with PLG drops. At Segment 1 end, four patients showed regression, proceeded to Segment 2, and treated with PLG drops for 8 weeks to prevent recurrence; seven patients had residual hard pseudomembranes and underwent surgical removal followed by 8 weeks with PLG. At Segment 2 end, 10 patients compliant per protocol without recurrence, transitioned to Segment 3. The primary endpoint of no pseudomembrane recurrence was 100% in all patients compliant with the treatment regimen at end Segment 2. Regression of surface area of existing pseudomembranes at end Segment 1 was achieved in 83.3% of affected eyes. Prior to trial enrollment, all patients experienced recurrences with prior historical therapies.
Eleven patients were treated in segment 3 with 3 ongoing (as of July, 2020). Eight patients exited the study: one patient due to noncompliance, and two who developed systemic manifestations requiring other therapy. No other subjects have experienced new eye lesions.
No major safety concerns arose during any the 3 Segments. Final data on immunogenicity will be available at the end of 2020.
CONCLUSIONS
Kedrion PLG eye drops were well tolerated and effective in recurrence prevention of pseudomembranes in patients with LC due to PLG deficiency who were compliant with the study protocol with long term follow-up now at 7 years (median: 74 months, range: 37-85 months); 3 subjects are still ongoing.
Caputo:Kedrion SpA: Research Funding. Suffredini:Kedrion SpA: Current Employment. Calcinai:Kedrion SpA: Current Employment. Crea:Kedrion SpA: Current Employment. Mathew:Kedrion SpA: Consultancy, Membership on an entity's Board of Directors or advisory committees. Nakar:Kedrion SpA: Research Funding; Prometic Biotherapeutics: Research Funding. Shapiro:Genentech/Roche: Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Kedrion Biopharma: Research Funding; Daiichi Sankyo: Research Funding; BioMarin: Research Funding; Novartis: Research Funding; Novo Nordisk: Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Agios: Research Funding; ProMetic Bio Therapeutics: Consultancy, Research Funding; OPKO: Research Funding; Glover Blood Therapeutics: Research Funding; Sigilon: Consultancy, Membership on an entity's Board of Directors or advisory committees; Takeda: Membership on an entity's Board of Directors or advisory committees, Research Funding; Novo Nordisk Hemophilia Foundation: Membership on an entity's Board of Directors or advisory committees; Catalyst BioSciences: Membership on an entity's Board of Directors or advisory committees; Bioverativ: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Pfizer: Research Funding; Octapharma: Research Funding; Sangamo: Research Funding.
Plasminogen eye drops, for treatment of Ligneous conjunctivitis due to Plasminogen Type I deficiency
Author notes
Asterisk with author names denotes non-ASH members.

This icon denotes a clinically relevant abstract

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal